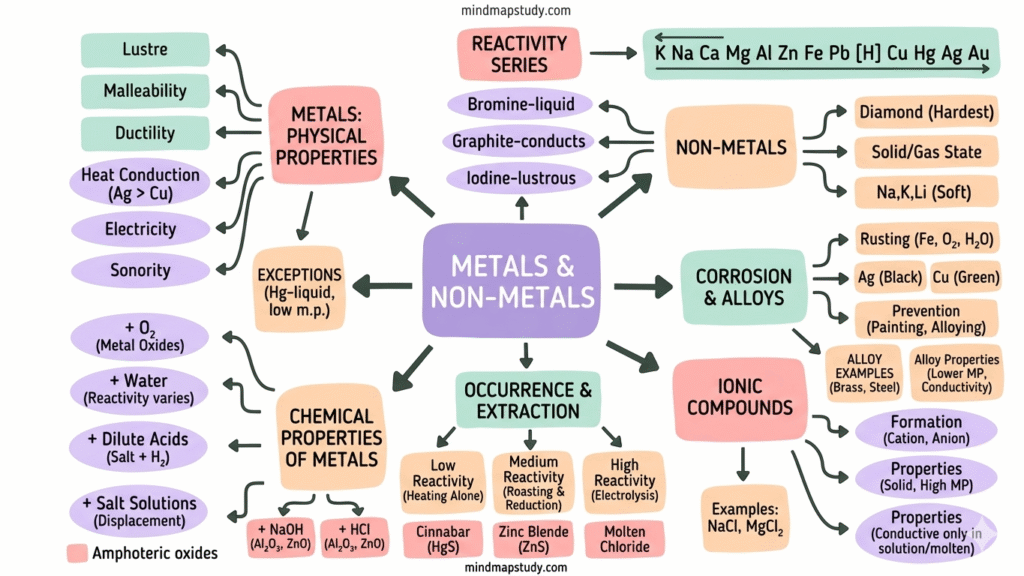
⚗️ Metals and Non-metals
Easy-to-understand notes with solved questions for school students
Table of Contents
Toggle3.1 Physical Properties
🔩 3.1.1 Metals — Physical Properties
✨ 1. Lustre (Shiny Surface)
Pure metals have a shining surface called metallic lustre. When you rub a metal piece with sandpaper, it looks shiny and bright.
🔨 2. Malleability (Can be beaten into sheets)
Metals can be beaten into thin, flat sheets without breaking. This property is called malleability.
🧵 3. Ductility (Can be drawn into wires)
The ability of a metal to be pulled into thin wires is called ductility. Gold is the most ductile metal — 1 gram of gold can be drawn into a 2 km long wire!
🌡️ 4. Good Conductors of Heat
Metals allow heat to pass through them easily. Silver and copper are the best conductors of heat. Lead and mercury are poor conductors.
⚡ 5. Good Conductors of Electricity
Metals allow electric current to pass through them. That is why electrical wires are made of copper or aluminium. The coating on wires (PVC/rubber) is a non-metal and does not conduct electricity.
🔔 6. Sonorous (Makes a ringing sound)
Metals produce a ringing sound when struck. That is why school bells and musical instruments are made of metals.
- Mercury is the only metal that is liquid at room temperature.
- Gallium & Caesium melt in the palm of your hand (very low melting points).
- Iodine is a non-metal but it is lustrous (shiny).
- Graphite (carbon) is a non-metal that conducts electricity.
- Diamond (carbon) is the hardest natural substance known.
- Sodium, Potassium, Lithium are so soft they can be cut with a knife.
🌿 3.1.2 Non-metals — Physical Properties
Non-metals are generally the opposite of metals. They can be solids or gases. Bromine is the only liquid non-metal at room temperature. Examples: carbon, sulphur, iodine, oxygen, hydrogen.
| Property | Metals | Non-metals |
|---|---|---|
| Lustre | Shiny | Dull (except iodine) |
| Hardness | Generally hard | Generally soft (diamond is exception) |
| Malleability | Malleable | Brittle (break on hammering) |
| Ductility | Ductile | Not ductile |
| Heat Conduction | Good conductors | Poor conductors |
| Electricity | Good conductors | Poor conductors (graphite exception) |
| Sonorous | Yes | No |
| State at room temp | Solid (except Hg) | Solid/Gas (except Br which is liquid) |
❓ Questions (Section 3.1)
Q1. Give an example of a metal which:
(i) is a liquid at room temperature
(ii) can be easily cut with a knife
(iii) is the best conductor of heat
(iv) is a poor conductor of heat
Q2. Explain the meanings of malleable and ductile.
Ductile: A metal that can be drawn into thin wires without breaking is called ductile. Example: Gold (most ductile).
3.2 Chemical Properties of Metals
🔥 3.2.1 Metals + Oxygen (Burning in Air)
Almost all metals combine with oxygen to form metal oxides. Metal oxides are basic in nature.
For example:
Oxides of sodium and potassium dissolve in water to form alkalis:
- K, Na — react so vigorously they catch fire; stored in kerosene
- Mg, Al, Zn, Pb — surface is covered by a protective oxide layer
- Iron (Fe) — iron filings burn vigorously in burner flame
- Cu — does not burn; forms black copper(II) oxide layer
- Ag, Au — do not react with oxygen even at high temperatures
Anodising is a process where aluminium is made the anode in an electrolytic cell with dilute sulphuric acid. Oxygen released at the anode forms a thick, protective oxide layer on aluminium that is resistant to corrosion and can also be dyed in attractive colours.
💧 3.2.2 Metals + Water
| Metal | Reacts with | Equation |
|---|---|---|
| K (Potassium) | Cold water (violently) | 2K + 2H₂O → 2KOH + H₂↑ + Heat |
| Na (Sodium) | Cold water (violently) | 2Na + 2H₂O → 2NaOH + H₂↑ + Heat |
| Ca (Calcium) | Cold water (less violent) | Ca + 2H₂O → Ca(OH)₂ + H₂↑ |
| Mg (Magnesium) | Hot water | Mg + 2H₂O → Mg(OH)₂ + H₂↑ |
| Al, Fe, Zn | Steam only | 2Al + 3H₂O → Al₂O₃ + 3H₂↑ 3Fe + 4H₂O → Fe₃O₄ + 4H₂↑ |
| Pb, Cu, Ag, Au | Does NOT react | — |
Note: Calcium and Magnesium float on water because hydrogen bubbles stick to their surface.
🧪 3.2.3 Metals + Dilute Acids
Reactivity order with dilute HCl: Mg > Al > Zn > Fe. Copper does not react with dilute HCl at all.
Aqua Regia = Concentrated HCl : Concentrated HNO₃ in ratio 3:1. It can dissolve even gold and platinum — metals that cannot be dissolved by either acid alone. The name means "Royal Water" in Latin.
⚗️ 3.2.4 Metals + Solutions of other Metal Salts
A more reactive metal displaces a less reactive metal from its salt solution. This is called a displacement reaction.
Example: Iron is more reactive than copper, so:
The blue colour of copper sulphate solution fades and a reddish-brown copper deposit forms on the iron nail.
3.2.5 The Reactivity Series
Metals are arranged from most reactive to least reactive in the Activity Series.
❓ Questions (Section 3.2)
Q1. Why is sodium kept immersed in kerosene oil?
Q2. Write equations for the reactions of:
(i) iron with steam
(ii) calcium and potassium with water
2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g) + Heat
Q3. Metals A, B, C, D and their reactions:
| Metal | FeSO₄ | CuSO₄ | ZnSO₄ | AgNO₃ |
|---|---|---|---|---|
| A | No reaction | Displacement | — | — |
| B | Displacement | — | No reaction | — |
| C | No reaction | No reaction | No reaction | Displacement |
| D | No reaction | No reaction | No reaction | No reaction |
(i) Which is the most reactive metal?
(ii) What would you observe if B is added to CuSO₄ solution?
(iii) Arrange metals A, B, C and D in decreasing order of reactivity.
Q4. Which gas is produced when dilute HCl is added to a reactive metal? Write the reaction of iron with dilute H₂SO₄.
Fe(s) + H₂SO₄(aq) → FeSO₄(aq) + H₂(g)↑
Q5. What happens when zinc is added to FeSO₄ solution?
Zn(s) + FeSO₄(aq) → ZnSO₄(aq) + Fe(s)
3.3 How Metals & Non-metals React (Ionic Bonds)
Metals tend to lose electrons to get a stable electron configuration (like noble gases). Non-metals tend to gain electrons. When a metal gives electrons to a non-metal, an ionic (electrovalent) compound is formed.
🔬 Formation of Sodium Chloride (NaCl)
Cl (2,8,7) → gains 1 electron → Cl⁻ (2,8,8)
Na⁺ and Cl⁻ attract each other → NaCl is formed
🔬 Formation of Magnesium Chloride (MgCl₂)
Each Cl (2,8,7) gains 1 electron → Cl⁻ (2,8,8)
1 Mg²⁺ + 2 Cl⁻ → MgCl₂ is formed
🧱 3.3.1 Properties of Ionic Compounds
| Property | Description |
|---|---|
| Physical state | Solid; hard but brittle (break under pressure) |
| Melting & Boiling point | Very high (strong electrostatic forces need lots of energy to break) |
| Solubility | Generally soluble in water; insoluble in kerosene, petrol |
| Electrical conductivity | Conduct electricity in molten state and in aqueous solution; NOT in solid state |
| Ionic Compound | Melting Point (K) | Boiling Point (K) |
|---|---|---|
| NaCl | 1074 | 1686 |
| LiCl | 887 | 1600 |
| CaCl₂ | 1045 | 1900 |
| CaO | 2850 | 3120 |
| MgCl₂ | 981 | 1685 |
❓ Questions (Section 3.3)
Q1(i). Write electron-dot structures for sodium, oxygen and magnesium.
Magnesium (Mg): 2 electrons in outermost shell → ·Mg·
Oxygen (O): 6 electrons in outermost shell → has 2 lone pairs + 2 unpaired electrons
Q1(ii). Show formation of Na₂O and MgO by electron transfer.
MgO: Mg loses 2 electrons → Mg²⁺. Oxygen gains 2 electrons → O²⁻. Combined: MgO.
Q1(iii). What ions are present in Na₂O and MgO?
Q2. Why do ionic compounds have high melting points?
3.4 Occurrence & Extraction of Metals
🌍 Where Do Metals Come From?
The Earth's crust is the main source of metals. Natural substances containing metals are called minerals. Minerals from which metals can be profitably extracted are called ores. The unwanted impurities in an ore are called gangue.
- Low reactivity metals (Au, Ag, Pt, Cu) → Found in FREE (native) state
- Medium reactivity metals (Zn, Fe, Pb, Cu) → Found as oxides, sulphides or carbonates
- High reactivity metals (K, Na, Ca, Mg, Al) → Never found free; always in compounds
⬇️ 3.4.3 Metals Low in Activity Series (Heating alone)
These metals are least reactive. Their oxides break down just by heating.
⬛ 3.4.4 Metals in the Middle — Roasting & Calcination
Roasting: Sulphide ores heated strongly in excess air → converts to oxide.
Calcination: Carbonate ores heated in limited air → converts to oxide.
The metal oxide is then reduced by carbon (coke):
Fe₂O₃(s) + 2Al(s) → 2Fe(l) + Al₂O₃(s) + Heat
⬆️ 3.4.5 Metals at the Top — Electrolysis
Highly reactive metals (Na, Mg, Ca, Al) cannot be reduced by carbon — they have more affinity for oxygen than carbon. They are extracted by electrolytic reduction of their molten chlorides/oxides.
🔋 3.4.6 Electrolytic Refining
Impure metals are purified by electrolysis:
- Anode = Impure metal (dissolves)
- Cathode = Thin strip of pure metal (pure metal deposits here)
- Electrolyte = Solution of the metal's salt
- Anode mud = Insoluble impurities that settle below the anode
❓ Questions (Section 3.4)
Q1. Define: (i) Mineral (ii) Ore (iii) Gangue
Ore: A mineral from which a particular metal can be extracted profitably is called an ore.
Gangue: The unwanted impurities (like soil, sand, etc.) mixed with the ore are called gangue.
Q2. Name two metals found in nature in the free state.
Q3. What chemical process is used for obtaining a metal from its oxide?
3.5 Corrosion & Alloys
🦀 What is Corrosion?
When metals are exposed to moist air over time, their surface slowly gets eaten away. This is called corrosion.
| Metal | What happens? | Product formed |
|---|---|---|
| Silver (Ag) | Reacts with sulphur in air | Black silver sulphide (Ag₂S) |
| Copper (Cu) | Reacts with moist CO₂ in air | Green basic copper carbonate |
| Iron (Fe) | Reacts with moist air (O₂ + H₂O) | Brown flaky rust (Fe₂O₃·xH₂O) |
🛡️ 3.5.1 Prevention of Corrosion
- Painting / Oiling / Greasing — creates a barrier between metal and air/water
- Galvanisation — coating iron/steel with a thin layer of zinc. Even if zinc coating breaks, it protects iron because zinc is more reactive and corrodes first.
- Chrome plating / Anodising
- Alloying — mixing metal with another substance to change its properties
🔀 Alloys
An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal. Alloys generally have lower electrical conductivity and lower melting point than the pure metals.
| Alloy | Composition | Properties / Uses |
|---|---|---|
| Steel | Fe + C (0.05%) | Hard and strong; used in construction |
| Stainless Steel | Fe + Ni + Cr | Hard, does not rust; utensils, cutlery |
| Brass | Cu + Zn | Not a good conductor; decorative items |
| Bronze | Cu + Sn | Not a good conductor; statues, medals |
| Solder | Pb + Sn | Low melting point; welding wires |
| Amalgam | Metal + Mercury (Hg) | Special uses (dental fillings) |
| 22-Carat Gold | Au (22 parts) + Cu or Ag (2 parts) | Used in jewellery (pure gold is too soft) |
The famous iron pillar near the Qutub Minar in Delhi is over 1600 years old and 8 metres tall, weighing 6 tonnes. It has NOT rusted! Ancient Indian metallurgists developed a special technique to protect iron from corrosion — a wonder even scientists today admire.
❓ Questions (Section 3.5)
Q1. In which cases will displacement reactions take place (ZnO, MgO, CuO heated with Zn, Mg, Cu)?
✅ Mg + ZnO → MgO + Zn (reaction occurs)
✅ Mg + CuO → MgO + Cu (reaction occurs)
✅ Zn + CuO → ZnO + Cu (reaction occurs)
❌ Zn + MgO → No reaction (Zn less reactive than Mg)
❌ Cu + ZnO → No reaction
❌ Cu + MgO → No reaction
Q2. Which metals do not corrode easily?
Q3. What are alloys?
✅ Solved Exercises
Q1. Which pair will give a displacement reaction?
(b) MgCl₂ + Al → No reaction (Al less reactive than Mg)
(c) FeSO₄ + Ag → No reaction (Ag less reactive than Fe)
✅ (d) AgNO₃ + Cu → Cu displaces Ag
Cu(s) + 2AgNO₃(aq) → Cu(NO₃)₂(aq) + 2Ag(s)
Q2. Best method to prevent an iron frying pan from rusting?
Q3. An element forms a high-melting compound with oxygen that is also soluble in water. The element is likely to be:
Q4. Food cans are coated with tin and not zinc because:
Q5. How can a hammer, battery, bulb, wires and switch help distinguish metals from non-metals?
Using the electrical circuit: Connect the sample between the circuit's terminals. If the bulb glows, it is a metal (conductor). If not, it is a non-metal (insulator).
(b) Usefulness: These tests are helpful but not perfect — graphite (non-metal) conducts electricity, and sodium (metal) is too soft to hammer. So both tests together are more reliable.
Q6. What are amphoteric oxides? Give two examples.
Examples: Al₂O₃ (aluminium oxide) and ZnO (zinc oxide).
Q7. Two metals that displace hydrogen from dilute acids, and two that do not.
Do NOT displace H₂: Copper (Cu), Silver (Ag)
Q8. In electrolytic refining of metal M, what is the anode, cathode, and electrolyte?
Cathode: Thin strip of pure metal M
Electrolyte: Aqueous solution of a salt of metal M (e.g., for copper: acidified CuSO₄)
Q9. Pratyush heats sulphur and collects the gas in an inverted test tube. (a) Action of gas on litmus paper? (b) Write the balanced equation.
(a)(i) Dry litmus paper: No change — SO₂ alone does not affect dry litmus (it needs water to form acid).
(a)(ii) Moist litmus paper: Turns red — SO₂ dissolves in water to form sulphurous acid (H₂SO₃), which is acidic.
(b) S(s) + O₂(g) → SO₂(g)
Q10. State two ways to prevent rusting of iron.
2. Painting or oiling — creating a physical barrier between iron and moist air.
Q11. What type of oxides are formed when non-metals combine with oxygen?
Q12. Give reasons:
(a) Platinum, gold and silver are used to make jewellery.
(b) Na, K and Li are stored under oil.
(c) Aluminium is highly reactive yet used for cooking utensils.
(d) Carbonate and sulphide ores are converted into oxides before extraction.
Q13. Why does lemon/tamarind juice clean tarnished copper vessels?
Q14. Differentiate between metals and non-metals based on chemical properties.
| Property | Metals | Non-metals |
|---|---|---|
| Reaction with O₂ | Form basic oxides | Form acidic oxides |
| Ion formation | Lose electrons → positive ions (cations) | Gain electrons → negative ions (anions) |
| React with acids | Displace H₂ from dilute acids | Do not react with dilute acids |
| React with H₂ | Generally do not form hydrides | Form hydrides (e.g., HCl, H₂S) |
| Displacement | More reactive displaces less reactive | Cannot displace H₂ from acids |
Q15. A goldsmith dipped gold bangles in a solution — they sparkled but became lighter. What solution did he use?
Q16. Why is copper used for hot water tanks and not steel (iron alloy)?
📝 Quick Revision — What You Learnt
- Metals are lustrous, malleable, ductile and good conductors of heat and electricity; solids at room temperature (except mercury).
- Non-metals form acidic oxides; metals form basic oxides. Amphoteric oxides (Al₂O₃, ZnO) react with both.
- The Activity Series lists metals in decreasing order of reactivity: K > Na > Ca > Mg > Al > Zn > Fe > Pb > [H] > Cu > Hg > Ag > Au.
- Ionic compounds are formed when metals transfer electrons to non-metals; they have high melting points and conduct electricity only in molten/dissolved state.
- Metals occur as free elements or in compounds (ores). Metallurgy = extraction + refining.
- Corrosion (e.g., rusting) needs both oxygen AND water. Prevention: painting, galvanising, alloying.
- An alloy is a homogeneous mixture of a metal with another metal or non-metal.
Download Free Mind Map PDF
This mind map contains all important topics of this chapter
Visit our Class 10 Science page for free mind maps of all Chapters
